DSHS distributes additional cases of remdesivir in Austin, surrounding area
AUSTIN, Texas - The Texas Department of State Health Services (DSHS) is distributing 13 additional cases of the antiviral drug remdesivir to eight hospitals across Central Texas. This will bring the total number of cases distributed by DSHS to Texas hospitals to 484.
Counties included in this distribution are Travis and Williamson.
Last week, DSHS distributed 10 initial cases across the Austin area.
SIGN UP FOR FOX 7 AUSTIN EMAIL ALERTS
"The Lone Star State continues to prioritize the health of our fellow Texans and ensure that medical providers across the state have the supplies needed to respond to COVID-19," said Gov. Greg Abbott. "I am grateful to our partners at the U.S. Department of Health and Human Services for providing these cases of remdesivir to communities across the state of Texas. Texas will continue to act swiftly to provide for our hospitals, secure treatment for patients, and slow the spread of COVID-19."
RELATED: Abbott, Texas DSHS distribute antiviral drug in Austin to treat COVID-19
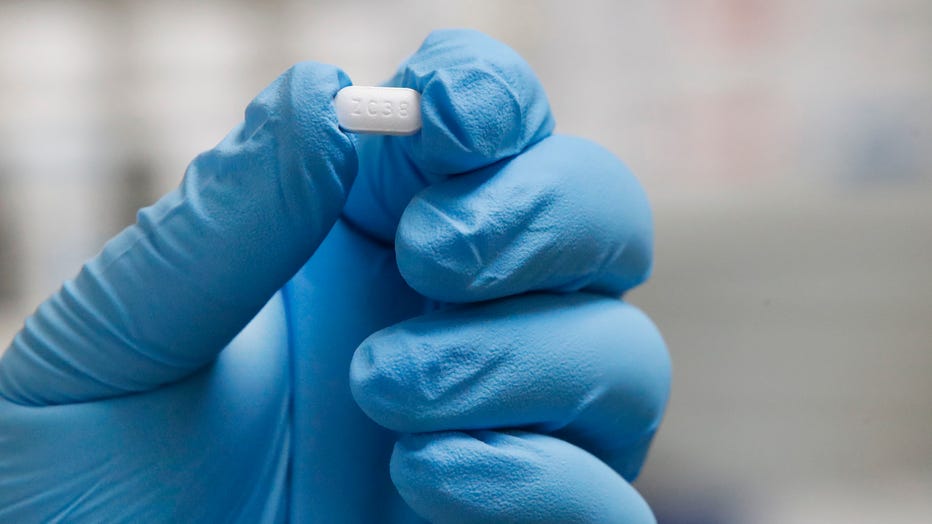
Remdesivir has shown promise in early trials in speeding up the recovery time among hospitalized COVID-19 patients, according to a press release provided by Abbott. The medication is being distributed according to COVID hospitalizations at the county level to ensure it is getting to where it is most needed. Additionally, the powdered formulation allows for the treatment of pediatric patients, so children's hospitals are included in the distribution.
---------
Get breaking news alerts in the FOX 7 Austin News app. It is FREE!
---------
DSHS reviewed the distribution methodology with the Infectious Disease Task Force Rapid Assessment Subcommittee, Texas Hospital Association, and the chair of the Texas Health Care Industry Strike Force on COVID-19. All three groups were supportive of the methodology as proposed with the data available.
Medical staff at each hospital will determine how the drug will be used, though it must be prescribed in accordance with the Food and Drug Administration's Emergency Use Authorization, allowing for the treatment of suspected or confirmed COVID-19 in adults and children hospitalized with severe disease, such as those in intensive care.
RELATED: FDA allows emergency use of remdesivir drug for coronavirus
Preliminary results from a clinical trial showed the average recovery time among patients who received remdesivir was 11 days versus 15 days with a placebo.
The supply is part of a donation from drug maker Gilead.
CLICK HERE FOR THE LATEST INFO ON THE CORONAVIRUS OUTBREAK
---
FOX 7 Austin is working to keep you up to date with coronavirus, with both local and national developments. Every weekday we're live at 12 p.m. with a special show reporting the latest news, prevention tips and treatment information.
You can watch live in your FOX 7 Austin app or on the FOX 7 Austin Facebook page.
You can also get the latest COVID-19 news from around the country at coronavirusnow.com.

