FDA approves Gilead's remdesivir, first drug for treating COVID-19
WASHINGTON - The U.S. Food and Drug Administration on Thursday approved Gilead Sciences’ antiviral drug remdesivir as a treatment for COVID-19 patients requiring hospitalization.
The drug is now the first fully approved treatment in the U.S. for the novel coronavirus, which has killed more than 1.1 million people around the world, according to Johns Hopkins University.
Remdesivir, which is sold under the brand name Veklury, was granted an emergency use authorization by the FDA in May.
A large study led by the U.S. National Institutes of Health found the drug cut the time to recovery by five days — from 15 days to 10 on average. It is given to patients through an IV, and in some cases, also a steroid.
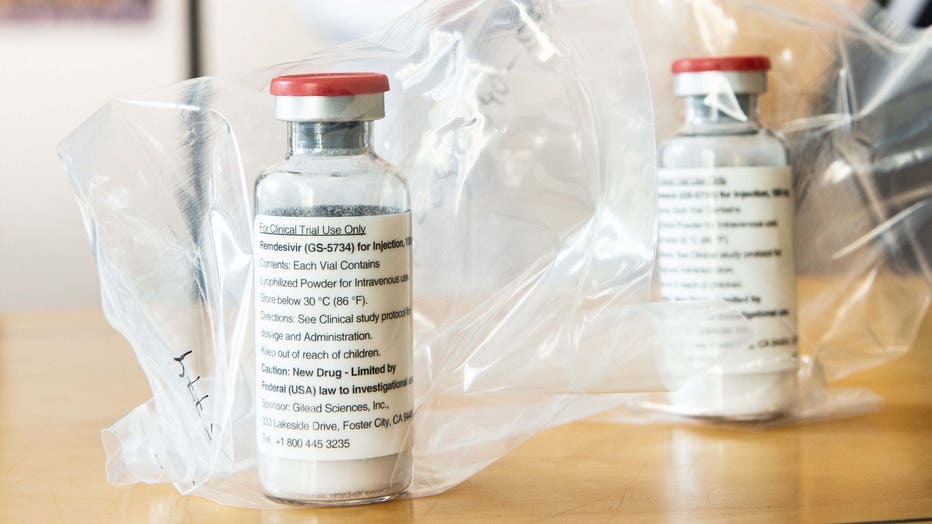
Vials of the drug Remdesivir are pictured during a press conference at the University Hospital Eppendorf (UKE) in Hamburg, Germany on April 8, 2020, amidst the COVID-19 pandemic. (Photo by ULRICH PERREY/POOL/AFP via Getty Images)
The California-based Gilead Sciences said Veklury is approved for people at least 12 years old and weighing at least 88 pounds who need hospitalization for their virus infection. It works by inhibiting a substance the virus uses to make copies of itself.
“Since the beginning of the COVID-19 pandemic, Gilead has worked relentlessly to help find solutions to this global health crisis,” Gilead CEO Daniel O’Day said in a statement. “It is incredible to be in the position today, less than one year since the earliest case reports of the disease now known as COVID-19, of having an FDA-approved treatment in the U.S. that is available for all appropriate patients in need.”
The drug is also approved for use against COVID-19 in the United Kingdom and Europe, and is among the treatments U.S. President Donald Trump received when he was infected earlier this month.
This story was reported from Cincinnati. The Associated Press contributed.


