Glioblastoma vaccine shows early promise in boosting survival

Trial participant Kim Garland (left) reviews a scan with the study’s primary investigator, Tanner Johanns, MD, PhD, a WashU Medicine oncologist. (Courtesy: Scott Garland)
A personalized vaccine for glioblastoma, an aggressive and incurable brain cancer, appears safe and may extend survival in some patients, according to an early-stage clinical trial co-led by researchers at Washington University School of Medicine in St. Louis.
Why you should care:
The experimental treatment triggered strong and broad immune responses and was linked to longer recurrence-free survival after surgery in a subset of patients. Among those with particularly aggressive disease, the vaccine caused no serious side effects and was associated with improved overall survival compared with historical outcomes following standard surgery and chemo-radiotherapy. One patient remains cancer-free nearly five years after diagnosis.
RELATED: ‘Miracle fruit’ may help cancer patients fight chemo-related weight loss
The findings come from a phase 1 trial conducted at Siteman Cancer Center at Barnes-Jewish Hospital and WashU Medicine and were published May 12 in Nature Cancer. The study was led in collaboration with Mass General Brigham and Geneos Therapeutics, a Philadelphia-based biotechnology company.
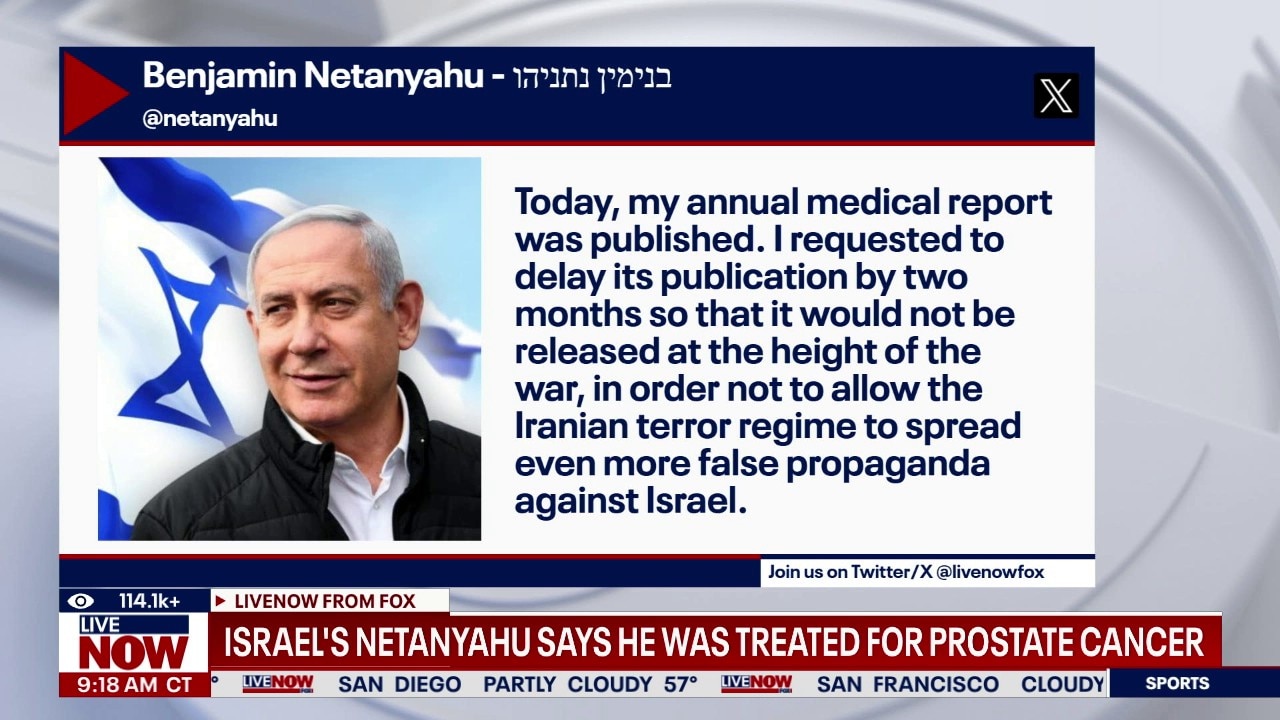
Israeli PM Netanyahu reveals cancer diagnosis
Israeli Prime Minister Benjamin Netanyahu on Friday revealed he had undergone treatment for prostate cancer and delayed releasing the results at the peak of U.S. and Israeli military operations against Iran earlier this year.
What they're saying:
"We are extremely encouraged by these results," Tanner M. Johanns, MD, PhD, lead author, said in a news release. "This kind of vaccine is a first for glioblastoma, and it is exciting to think how we can leverage this individualized therapeutic DNA cancer vaccine platform to make a positive impact on the lives of patients who are fighting this disease. Additionally, combination therapies leveraging this personalized platform are currently being investigated at WashU to test if outcomes may be improved further."
RELATED: New drug significantly extends survival in advanced pancreatic cancer trial
Dig deeper:
The treatment uses engineered DNA to train the immune system to attack the cancer. Because each tumor carries its own unique proteins, the vaccine is tailored to help the body recognize and destroy those specific cancer cells.
While some immunotherapies for glioblastoma have shown promise, the cancer often adapts and evades the immune system. This vaccine is designed to target many different proteins at once, making it harder for the tumor to escape.
RELATED: New drug significantly extends survival in advanced pancreatic cancer trial
Glioblastoma is also considered a "cold" tumor, meaning it can hide from immune defenses. The vaccine aims to turn it into a "hot" tumor, making it more visible and vulnerable to attack.
In the study, the approach helped the immune system target up to 40 tumor-specific proteins per patient — about twice as many as previous cancer vaccines.
The backstory:
The vaccine, called GNOS-PV01, targets "neoantigens" — proteins found only on an individual patient’s cancer cells. Researchers used a computer algorithm to identify these proteins from different parts of each patient’s tumor, helping the vaccine attack a wider range of targets.
The idea builds on earlier DNA-based vaccine work in breast cancer and was adapted for glioblastoma. In the trial, nine newly diagnosed patients were treated at Siteman Cancer Center. Each patient received a custom-made vaccine based on the genetic profile of their tumor.
RELATED: Pig semen eye drops may treat rare childhood eye cancer, study says
Shots began about 10 weeks after surgery and were given regularly over several months. All but one patient showed increased immune activity in response to the treatment.
The early results were encouraging: about two-thirds of patients had no cancer progression six months after surgery, and the same proportion were alive at one year — higher than typical rates. One-third were still alive after two years, roughly double historical outcomes, and one patient remains cancer-free nearly five years after diagnosis.
The Source: The information in this story comes from a phase 1 clinical trial conducted at Siteman Cancer Center at Barnes-Jewish Hospital and Washington University School of Medicine in St. Louis, with collaboration from Mass General Brigham and Geneos Therapeutics. This story was reported from Los Angeles.

